Uranium Enrichment
- Most of the about 500 commercial nuclear power reactors operating or under construction in the world today require uranium 'enriched' in the U-235 isotope for their fuel.
- The commercial process employed for this enrichment involves gaseous uranium in centrifuges. A process based on laser excitation is under development.
- Prior to enrichment, uranium oxide must be converted to a fluoride so that it can be processed as a gas, at low temperature.
- From a non-proliferation standpoint, uranium enrichment is a sensitive technology needing to be subject to tight international control.
- In recent years there has been a significant surplus of world enrichment capacity.
Uranium found in nature consists largely of two isotopes, U-235 and U-238. The production of energy in nuclear reactors is from the 'fission' or splitting of the U-235 atoms, a process which releases energy in the form of heat. U-235 is the main fissile isotope of uranium.
Natural uranium contains 0.7% of the U-235 isotope. The remaining 99.3% is mostly the U-238 isotope which does not contribute directly to the fission process (though it does so indirectly by the formation of fissile isotopes of plutonium). Isotope separation is a physical process to concentrate (‘enrich’) one isotope relative to others. Most reactors are light water reactors (of two types – PWR and BWR) and require uranium to be enriched from 0.7% to 3-5% U-235 in their fuel. This is normal low-enriched uranium (LEU). There is some interest in taking enrichment levels to about 7%, and even close to 20% for certain special power reactor fuels, as high-assay LEU (HALEU).
See also information page on High-Assay Low-Enriched Uranium (HALEU).
Uranium-235 and U-238 are chemically identical, but differ in their physical properties, notably their mass. The nucleus of the U-235 atom contains 92 protons and 143 neutrons, giving an atomic mass of 235 units. The U-238 nucleus also has 92 protons but has 146 neutrons – three more than U-235 – and therefore has a mass of 238 units.
The 1.27% difference in mass between U-235 and U-238 allows the isotopes to be separated and makes it possible to increase or "enrich" the percentage of U-235. All present and historic enrichment processes, directly or indirectly, make use of this small mass difference.
Some reactors, for example the Canadian-designed Candu, use natural uranium as their fuel. (For comparison, uranium used for nuclear weapons would have to be enriched in plants specially designed to produce at least 90% U-235.)
Enrichment processes require uranium to be in a gaseous form at relatively low temperature, hence uranium oxide from the mine is converted to uranium hexafluoride in a preliminary process, at a separate conversion plant.
In recent years there has been a significant over-supply of enrichment capacity worldwide, much of which has been used to diminish uranium demand or supplement uranium supply. The ability of enrichment to substitute for uranium (see description of underfeeding below) has become more significant as centrifuge technology has taken over, since this means both lower SWU costs and the need to keep the centrifuges running, so capacity remains online even as demand drops away.
Enrichment capacity
Uranium enrichment is strategically sensitive and capital intensive, creating significant barriers to entry for any new supplier. Hence, there are relatively few commercial enrichment suppliers operating a limited number of facilities worldwide.
There are three major producers at present: Orano, Rosatom, and Urenco operating large commercial enrichment plants in France, Germany, Netherlands, UK, USA, and Russia. CNNC is a major domestic supplier and is pursuing export sales. In Japan and Brazil, domestic fuel cycle companies manage modest supply capability. Elsewhere, small non-safeguarded facilities are subject to international opposition.
World enrichment capacity – operational in 2022
| Operator | Capacity (thousand SWU/yr) | ||
|---|---|---|---|
| 2022 | 2025 | 2030 | |
| CNNC | 8900 | 10,000 | 17,000 |
| Orano | 7500 | 7500 | 7500 |
| Rosatom | 27,100 | 27,100 | 27,100 |
| Urenco | 17,900 | 17,900 | 17,900 |
| Other (INB, JNFL) | 100 | 400 | 800 |
| Total | 61,500 | 62,900 | 70,300 |
Source: World Nuclear Association Nuclear Fuel Report 2023 and company websites
Orano's SET subsidiary operates the Georges Besse II (GBII) gas centrifuge at Tricastin in the south of France. GBII was officially opened in December 2010 and commenced commercial operation in April 2011. At the end of 2016, GBII reached its planned production capacity of 7.5 million SWU/yr. GBII replaced Eurodif's Georges Besse gaseous diffusion plant, which was in operation at Tricastin since 1978. In October 2023, Orano decided to increase production capacity at the plant by 2.5 million SWU. A ceremony to mark the laying of the foundation stone took place on 10 October 2024.
In September 2024 Orano selected Oak Ridge as its preferred site for a new centrifuge enrichment facility. Orano's forerunner company Areva had previously planned to build a $2 billion, 3.3 million SWU/yr enrichment plant at Eagle Rock, Idaho Falls, USA. In 2009 it applied for doubling in capacity to 6.6 million SWU/yr. However, it was subsequently cancelled, and in 2018 Orano requested the NRC to terminate the licence.
Rosatom has four enrichment plants: Novouralsk, Zelenogorsk, Angarsk and Seversk. The four facilities have a combined capacity of over 27 million SWU/yr.
Urenco has enrichment operations in the UK (Capenhurst), Germany (Gronau) and the Netherlands (Almelo). In 2010 Urenco's US subsidiary Louisiana Energy Services commenced operation at its centrifuge plant in Eunice, New Mexico.
Urenco has announced a capacity programme to refurbish and potentially expand its centrifuges at all four of its sites to help meet the world’s increased demand for enrichment services. In July 2023, Urenco confirmed plans to add 700,000 SWU/yr new enrichment capacity at UUSA plant, with the first new cascades online in 2025. In December 2023 it approved an investment to expand its Almelo facility by 750,000 SWU/yr, with the first new cascades coming online around 2027.
CNNC's enrichment capacity is estimated at about 9 million SWU/yr, which includes 1.5 million SWU/yr from Russian-supplied centrifuges, with the remaining SWU capacity from indigenous technology. A production line using indigenous centrifuges was launched in March 2018 at the Shaanxi plant.
A small number of other countries have limited enrichment capability.
Argentina reactivated its gaseous diffusion plant at Pilcaniyeu in 2010, more than two decades after production there was halted. Production was again halted in 2018 and the plant has not been used for commercial or export needs. Argentina is also exploring the development of new centrifuge technology as well as laser enrichment, but this is a long-term project without any clear timetable.
Since 2009 Brazil's Indústrias Nucleares do Brasil (INB) has produced small quantities of enriched uranium for domestic consumption using domestically developed centrifuges as part of wider activities covering much of the front end of the nuclear fuel cycle.
INB is renovating the Resende Nuclear Fuel Factory (FCN) in Rio de Janeiro. The target of the first phase (2000-2023), which was completed successfully, was to construct enrichment capacity sufficient to supply 70% of the requirements of the Angra 1 unit, whilst the aim of the second phase (2023-2037) is for FCN to supply 100% of enrichment needed for the Angra plant (i.e. units 1&2, plus the under construction unit 3).
Japan Nuclear Fuel Limited (JNFL) is upgrading centrifuge capacity at its Rokkasho facility. JNFL commenced commercial operation of the newer machinery in 2012 and has since then installed approximately 75,000 SWU/yr of capacity. The full planned 1.5 million SWU/yr of capacity was expected to be installed by 2022 and operational by 2027, but has been beset by delays. A further 375,000 SWU/yr is now expected to be in operation by 2027.
India, Pakistan and Iran have small centrifuge enrichment capabilities, but do not sell SWU commercially or export to the global market.
In 2022, the enrichment market was impacted by the Russian-Ukrainian conflict. Many utilities, particularly in Europe and the USA, tried to diversify their nuclear fuel supply or even cancel deliveries of fuel from Russia. In this context, in July 2023, Urenco confirmed plans to add 700,000 SWU/yr new enrichment capacity at the UUSA plant, with the first new cascades online in 2025. Orano is studying possibilities to expand its enrichment capacity.
Prospective suppliers
Centrus Energy Corporation (formerly USEC) was building its American Centrifuge Plant in Piketon, Ohio, on the same Portsmouth site where the DOE's experimental plant operated in the 1980s as the culmination of a major R&D programme. Operation from 2012 was envisaged, at a cost of $3.5 billion then estimated. It was designed to have an initial annual capacity of 3.8 million SWU. Authorization for enrichment up to 10% was sought – most enrichment plants operate up to 5% U-235 product, which is becoming a serious constraint as reactor fuel burnup increases. A demonstration cascade started up in September 2007 with about 20 prototype machines, and a lead cascade of commercial centrifuges started operation in March 2010. These are very large machines, 13 m tall, each with about 350 SWU/yr capacity and requiring regular maintenance. The whole project was largely halted in July 2009 pending further finance, although a demonstration cascade became operational in October 2013. It was licensed for 7 million SWU/yr enrichment up to 10% U-235, but operation ceased in February 2016.
In mid-2021 it was licensed to produce HALEU up to 20%. In November 2022, the DOE awarded Centrus $150 million of cost-shared funding to finish the cascade, complete final regulatory steps, begin operating the cascade, and produce up to 20 kg of HALEU by the end of 2023 – this was delivered in November 2023. It was then expected that production would continue in 2024 at a rate of 900 kg of HALEU per year. However in February 2024 it was announced that delays in the supply of 5B cylinders from the DOE would prevent the delivery of this amount of material in 2024.
Global Laser Enrichment (GLE), a business venture of Silex (51%) and Cameco (49%), has been developing a next-generation, laser-based technology for potential commercial application in enriching uranium, with an option for Cameco to attain a majority interest of up to 75% ownership. Laser uranium enrichment technology provides highly selective excitation of U-235 in UF6 and is expected to be more flexible and cost-effective than other technologies. For more information see Laser processes section below.
International Enrichment Centres, Multilateral approaches
Following proposals from the International Atomic Energy Agency (IAEA) and Russia, and in connection with the US-led Global Nuclear Energy Partnership (GNEP), there have been moves to establish international uranium enrichment centres. These are one kind of multilateral nuclear approach (MNA) called for by the IAEA. Part of the motivation for international centres is to bring all new enrichment capacity, and perhaps eventually all enrichment, under international control as a non-proliferation measure. Precisely what 'control' means remains to be defined, and will not be uniform in all situations. But having ownership and operation shared among a number of countries at least means that there is a level of international scrutiny which is unlikely in a strictly government-owned and -operated national facility.
The first of these international centres is the International Uranium Enrichment Centre (IUEC) established in 2007 by Rosatom at Angarsk in Siberia, with Kazakh, Armenian and Ukrainian equity so far. Since 2012 the IUEC has been supplying 60,000 SWU/yr to its shareholder State Concern Nuclear Fuel (Ukraine). The centre is open to other countries that meet the requirements of the Nuclear Non-Profiliferation Treaty (NPT). Separately a fuel bank – a small reserve of LEU for guaranteed supply to other countries at the IAEA's request – was created in 2010 at the site of the IUEC.
In some respects this is very similar to what pertained with the Eurodif set-up, where a single large enrichment plant in France with five owners (France – 60%, Italy, Spain, Belgium and Iran) was operated under IAEA safeguards by the host country without giving participants any access to the technology – simply some entitlement to share of the product, and even that was constrained in the case of Iran. The French Atomic Energy Commission proposed that the new Georges Besse II plant which replaced Eurodif should be open to international partnerships on a similar basis, and minor shares in the Areva (now Orano) subsidiary operating company Societe d'Enrichissement du Tricastin (SET) have been sold to GDF Suez (now Engie), a Japanese partnership, and Korea Hydro and Nuclear Power (KHNP) – total 12%.
The three-nation Urenco set-up is also similar though with more plants in different countries – UK, Netherlands and Germany – and here the technology is not available to host countries or accessible to other equity holders. Like Russia with the IUEC, Urenco (owned by the UK and Netherlands host governments plus E.On and RWE in Germany) has said that if its technology is used in international centres it would not be accessible. Its new plant is in the USA, where the host government has regulatory but not management control.
Feedstock and Separative Work Units
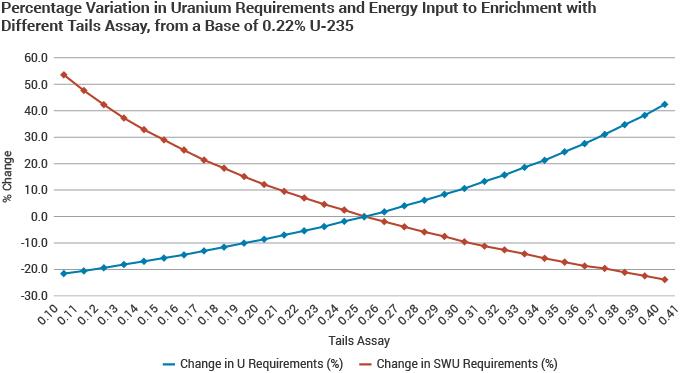
The feedstock for enrichment consists of uranium hexafluoride (UF6) from the conversion plant. Following enrichment two streams of UF6 are formed: the enriched ‘product’ containing a higher concentration of U-235 which will be used to make nuclear fuel, and the ‘tails’ containing a lower concentration of U-235, and known as depleted uranium (DU). The tails assay (concentration of U-235) is an important quantity since it indirectly determines the amount of work that needs to be done on a particular quantity of uranium in order to produce a given product assay. Feedstock may have a varying concentration of U-235, depending on the source. Natural uranium will have a U-235 concentration of approximately 0.7%, while recycled uranium will be around 1% and tails for re-enrichment often around 0.25-0.30%.
The capacity of enrichment plants is measured in terms of 'separative work units' or SWU. The SWU is a complex unit which indicates the energy input relative to the amount of uranium processed, the degree to which it is enriched (i.e. the extent of increase in the concentration of the U-235 isotope relative to the remainder) and the level of depletion of the remainder – called the ‘tails’. The unit is strictly: kilogram separative work unit, and it measures the quantity of separative work performed to enrich a given amount of uranium a certain amount when feed and product quantities are expressed in kilograms. The unit 'tonnes SWU' is also used.
For instance, to produce one kilogram of uranium enriched to 5% U-235 requires 7.9 SWU if the plant is operated at a tails assay 0.25%, or 8.9 SWU if the tails assay is 0.20% (thereby requiring only 9.4 kg instead of 10.4 kg of natural U feed). There is always a trade-off between the cost of enrichment SWU and the cost of uranium.
Today, 5% U-235 is the maximum level of enrichment for fuel used in normal power reactors. However, especially in relation to new small reactor designs, there is increasing interest in higher enrichment levels. The US Nuclear Regulatory Commission has approved a licence amendment for Urenco USA to enrich to 5.5% U-235, and it plans to apply for a 10% limit in 2023. Urenco's Capenhurst plant in the UK is licensed to enrich up to 6%. Global Nuclear Fuel-Americas, a GE Hitachi fuel fabricator, has applied to use 8% enriched uranium. Orano said in mid-2021 that it was preparing to go to 6% in 2023 and was planning for licence limits of 10%. The planned Wilmington North Carolina plant of Global Laser Enrichment (see below) is licensed to enrich to 8%. Rosatom has said that it can provide higher enrichment levels than 5%. Centrus in the USA was licensed in mid-2021 to produce up to 600 kg per year of high-assay LEU up to 20% in its small centrifuge plant.
High-assay LEU (HALEU) is anything between 5% and 20% U-235, and the higher levels in this range are needed for some advanced power reactor fuels. Some small demand already exists for research reactors. However such HALEU fuel is best produced onsite where it is converted from fluoride and fabricated into fuel, to avoid the need for special transport packages for HALEU UF6. For enrichment levels up to 10%, called LEU+, existing transport casks can be modified. Above 10% the HALEU requires enhanced physical security and different licensing.
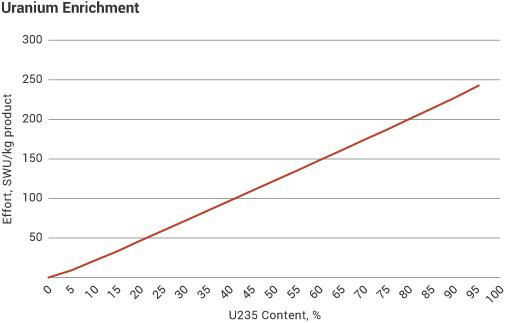
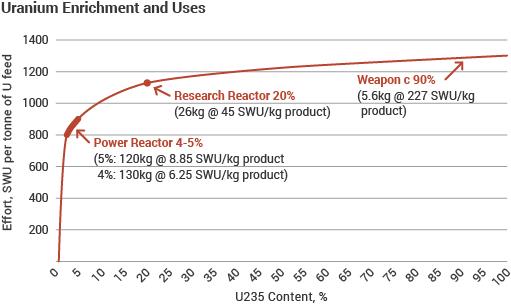
The first graph shows enrichment effort (SWU) per unit of product. The second shows how one tonne of natural uranium feed might end up: as 120-130 kg of uranium for power reactor fuel, as 26 kg of typical research reactor fuel, or conceivably as 5.6 kg of weapons-grade material. The curve flattens out so much because the mass of material being enriched progressively diminishes to these amounts, from the original one tonne, so requires less effort relative to what has already been applied to progress a lot further in percentage enrichment. The relatively small increment of effort needed to achieve the increase from normal levels is the reason why enrichment plants are considered a sensitive technology in relation to preventing weapons proliferation, and are very tightly supervised under international agreements. Where this safeguards supervision is compromised or obstructed, as in Iran, concerns arise.
About 140,000 SWU is required to enrich the annual fuel loading for a typical 1000 MWe light water reactor at today's higher enrichment levels. Enrichment costs are substantially related to electrical energy used. The gaseous diffusion process consumes about 2500 kWh (9000 MJ) per SWU, while modern gas centrifuge plants require only about 50 kWh (180 MJ) per SWU.
Enrichment accounts for almost half of the cost of nuclear fuel and about 5% of the total cost of the electricity generated. In the past it has also accounted for the main greenhouse gas impact from the nuclear fuel cycle where the electricity used for enrichment is generated from coal. However, it still only amounts to 0.1% of the carbon dioxide from equivalent coal-fired electricity generation if modern gas centrifuge plants are used.
The utilities which buy uranium from the mines need a fixed quantity of enriched uranium in order to fabricate the fuel to be loaded into their reactors. The quantity of uranium they must supply to the enrichment company is determined by the enrichment level required (% U-235) and the tails assay (also % U-235). This is the contracted or transactional tails assay, and determines how much natural uranium must be supplied to create a quantity of Enriched Uranium Product (EUP) – a lower tails assay means that more enrichment services (notably energy) are to be applied. The enricher, however, has some flexibility in respect to the operational tails assay at the plant. If the operational tails assay is lower than the contracted/transactional assay, the enricher can set aside some surplus natural uranium, which it is free to sell (either as natural uranium or as EUP) on its own account.
This is known as underfeeding.* Utilities are increasingly seeking to control this flexibility of operation in contracts, and themselves get some of the benefit from underfeeding.
* The opposite situation, where the operational tails assay is higher, requires the enricher to supplement the natural uranium supplied by the utility with some of its own – this is called overfeeding.
In respect to underfeeding (or overfeeding), the enricher will base its decision on the plant economics together with uranium and energy prices. The World Nuclear Association's 2021 Nuclear Fuel Report estimates that by underfeeding the enrichers have the potential to contribute more than 6000 tU/yr to world markets to 2025 on the basis of typical Western 0.22% tails assays, much of this potential in Russia, where tails assays are normally 0.10% U-235.
With reduced demand for enriched uranium following the Fukushima accident, enrichment plants have continued running, since it is costly to shut down and restart centrifuges. The surplus SWU output can be sold, or the plants can be underfed so that the enricher ends up with excess uranium for sale, or with enriched product for its own inventory and later sale. The inertia of the enrichment process thus exacerbates oversupply in the uranium market and depresses SWU prices (from $160/SWU in 2010, the spot price in March 2016 was $60, and $92 in March 2022).
Natural uranium is usually shipped to enrichment plants in type 48Y cylinders, each holding about 12.5 tonnes of uranium hexafluoride (8.4 tU). These cylinders are then used for long-term storage of DU, typically at the enrichment site. Enriched uranium is shipped in type 30B cylinders, each holding 2.27 t UF6 (1.54 tU).
Enrichment processes
A number of enrichment processes have been demonstrated historically or in the laboratory but only two, the gaseous diffusion process and the centrifuge process, have operated on a commercial scale. Centrifugation is the only enrichment process used today.
In both gaseous diffusion and centrifuge processes, UF6 gas is used as the feed material. Molecules of UF6 with U-235 atoms are about one percent lighter than the rest, and this difference in mass is the basis of both processes. Isotope separation is a physical process.*
*One chemical process has been demonstrated to pilot plant stage but not used. The French Chemex process exploited a very slight difference in the two isotopes' propensity to change valency in oxidation/reduction, utilising aqueous (III valency) and organic (IV) phases.
The various enrichment processes that have been used commercially or demonstrated in a laboratory are described below.
Centrifuge process
The gas centrifuge process was first demonstrated in the 1940s but was shelved in favour of the simpler diffusion process. It was then developed and brought on stream in the 1960s as the second-generation enrichment technology. It is economic on a smaller scale, e.g. under 2 million SWU/yr, which enables staged development of larger plants. It is much more energy efficient than diffusion, requiring only about 40-50 kWh per SWU.
All currently operating enrichment facilities use the centrifuge process.

A bank of centrifuges at a Urenco plant
Like the diffusion process, the centrifuge process uses UF6 gas as its feed and makes use of the slight difference in mass between U-235 and U-238. The gas is fed into a series of vacuum tubes, each containing a rotor 3 to 5 metres tall and 20 cm diameter.* European centrifuges produce 40-100 SWU/yr. When the rotors are spun rapidly, at 50,000 to 70,000 rpm, the heavier molecules with U-238 increase in concentration towards the cylinder's outer edge. There is a corresponding increase in concentration of U-235 molecules near the centre. The countercurrent flow set up by a thermal gradient enables enriched product to be drawn off axially, heavier molecules at one end and lighter ones at the other.
* USEC's American Centrifuges are more than 12 m tall and 40-50 cm diameter. The Russian centrifuges are less than one metre tall. Chinese ones are larger, but shorter than Urenco's.
The enriched gas forms part of the feed for the next stages while the depleted UF6 gas goes back to the previous stage. Eventually enriched and depleted uranium are drawn from the cascade at the desired assays.
To obtain efficient separation of the two isotopes, centrifuges rotate at very high speeds, with the outer wall of the spinning cylinder moving at between 400 and 500 metres per second to give a million times the acceleration of gravity.
Although the volume capacity of a single centrifuge is much smaller than that of a single diffusion stage, its capability to separate isotopes is much greater. Centrifuge stages normally consist of a large number of centrifuges in parallel. Such stages are then arranged in cascade similarly to those for diffusion. In the centrifuge process, however, the number of stages may only be 10 to 20, instead of a thousand or more for diffusion. Centrifuges are designed to run for about 25 years continuously, and cannot simply be slowed or shut down and restarted according to demand. Western cascades are designed for 0.18 to 0.22% tails assay, Russian ones for 0.10%.
Urenco Nederland uses centrifuge technology to produce a variety of stable isotopes for medical, industrial and research applications. Urenco Stable Isotopes has operated for over three decades at Almelo. A new cascade commissioned in 2021 is designed for the enrichment of multiple isotopes, including cadmium, germanium, iridium, molybdenum, selenium, tellurium, titanium, tungsten, xenon and zinc.
Laser processes
Laser enrichment processes have been the focus of interest for some time. They are a possible third-generation technology promising lower energy inputs, lower capital costs and lower tails assays, hence significant economic advantages. One of these processes is almost ready for commercial use. Laser processes are in two categories: atomic and molecular.
Development of the Atomic Vapour Laser Isotope Separation (AVLIS, and the French SILVA) began in the 1970s. In 1985 the US Government backed it as the new technology to replace its gaseous diffusion plants as they reached the end of their economic lives early in the 21st century. However, after some US$ 2 billion in R&D, it was abandoned in USA in favour of SILEX, a molecular process. French work on SILVA ceased following a 4-year program to 2003 to prove the scientific and technical feasibility of the process. Some 200kg of 2.5% enriched uranium was produced in this.
Atomic vapour processes work on the principle of photo-ionisation, whereby a powerful laser is used to ionize particular atoms present in a vapour of uranium metal. (An electron can be ejected from an atom by light of a certain frequency. The laser techniques for uranium use frequencies which are tuned to ionize a U-235 atom but not a U-238 atom.) The positively-charged U-235 ions are then attracted to a negatively-charged plate and collected. Atomic laser techniques may also separate plutonium isotopes.
Most molecular processes which have been researched work on a principle of photo-dissociation of UF6 to solid UF5+, using tuned laser radiation as above to break the molecular bond holding one of the six fluorine atoms to a U-235 atom. This then enables the ionized UF5 to be separated from the unaffected UF6 molecules containing U-238 atoms, hence achieving a separation of isotopes.* Any process using UF6 fits more readily within the conventional fuel cycle than the atomic process.
* A similar principle can be used in enriching atomic lithium, with magnetic separation of the ionized atoms, leaving pure Li-7.
The main molecular laser process to enrich uranium is SILEX, which utilises UF6 and is now known as Global Laser Enrichment (GLE). In 2006 GE Energy entered a partnership with Australia's Silex Systems to develop the third-generation SILEX process. It provided for GE (now GE Hitachi) to construct in the USA an engineering-scale test loop, then a pilot plant or lead cascade, before moving to a full commercial plant. GLE will now decide in the light of commercial considerations on whether to proceed with a full-scale enrichment facility at Wilmington. The project is licensed to enrich up to 8% U-235; enrichment to almost 20% U-235 is envisaged but not yet licensed.
As well as $20 million upfront and subsequent payments, the licence agreement with GE was to yield 7-12% perpetual royalties, the precise amount depending on the cost of deploying the commercial technology as well as several progress payments. In-mid 2008 Cameco bought into the GLE project, paying $124 million for a 24% share, alongside GE (51%) and Hitachi (25%). (Earlier, in 1996 USEC had secured the rights to evaluate and develop SILEX for uranium but bailed out of the project in 2003.) In April 2016 GE and Hitachi notified their intention to exit GLE, and during subsequent negotiations Silex funded 76% of GLE’s R&D at Wilmington, North Carolina. In February 2019 Silex Systems and Cameco agreed to buy out the GEH 76% share in GLE for US$ 20 million on a deferred payment basis, so that Cameco held 49% of GLE and Silex 51%. Cameco had an option to purchase an additional 26% of GLE. The agreement called for Silex and Cameco to pay $300,000 per month to complete construction of the prototype enrichment facility which had been partially built by GEH. Final US government approvals for the buyout were granted in January 2021. Silex said that “the Paducah commercial opportunity represents an ideal path to market for our disruptive SILEX laser enrichment technology.”
Earlier in August 2013 GLE submitted a proposal to the DOE to establish a “$1 billion” laser enrichment plant at Paducah, Kentucky to enrich high-assay tails (above 0.34% U-235) owned by the DOE to natural uranium level (0.7% U-235). There is about 115,000 tonnes of these at Paducah and Portsmouth (among a total of 550,000 t tails). Negotiations with the DOE continued into 2016, and in November an agreement was signed with the DOE for it to supply about 300,000 tonnes of high-assay tails, justifying construction by GLE of the Paducah Laser Enrichment Facility (PLEF). PLEF would become a commercial uranium enrichment production facility under a US NRC licence, producing about 100,000 tonnes of natural-grade uranium over 40 years or more. The DOE would dispose of the reduced-assay balance. The estimated plant size is 0.5 to 1.0 million SWU/yr, since purchases of DU may not exceed 2000 t/yr natural uranium equivalent.
Earlier in mid-2009 GEH submitted the last part of its licence application for this GLE plant at Wilmington, and following review of provisions for the physical protection of special nuclear material and classified matter, material control and accounting, plus further review by the NRC Atomic Safety and Licensing Board, a full licence to construct and operate a plant of up to 6 million SWU/yr was issued in September 2012.
Meanwhile, GLE is completing the test loop programme, the initial phase of which was successful in meeting performance criteria, and engineering design for a commercial facility commenced. GLE is operating the test loop at Global Nuclear Fuel's Wilmington, North Carolina fuel fabrication facility (GNF is a GE-led joint venture with Toshiba, and Hitachi). In October 2021 the first full-scale laser system module successfully completed initial testing at Silex’s Lucas Heights facility in Sydney.
GLE is planning to complete the commercial pilot demonstration project by the mid-2020s, after which a feasibility assessment will be conducted for the proposed Paducah Laser Enrichment Facility (PLEF), which GLE hopes to deploy for the production of natural grade uranium by 2030.
Applications to silicon and zirconium stable isotopes are also being developed by Silex Systems near Sydney.
CRISLA is another molecular laser isotope separation process which is the early stages of development. In this a gas is irradiated with a laser at a particular wavelength that would excite only the U-235 isotope. The entire gas is subjected to low temperatures sufficient to cause condensation on a cold surface or coagulation in the un-ionized gas. The excited molecules in the gas are not as likely to condense as the unexcited molecules. Hence in cold-wall condensation, gas drawn out of the system is enriched in the U-235 isotope that was laser-excited. NeuTrek, the development company, is aiming to build a pilot plant in USA.
Gaseous diffusion process
The energy-intensive gaseous diffusion process of uranium enrichment is no longer used in the nuclear industry. It involves forcing uranium hexafluoride gas under pressure through a series of porous membranes or diaphragms. As U-235 molecules are lighter than the U-238 molecules they move faster and have a slightly better chance of passing through the pores in the membrane. The UF6 which diffuses through the membrane is thus slightly enriched, while the gas which did not pass through is depleted in U-235.
This process is repeated many times in a series of diffusion stages called a cascade. Each stage consists of a compressor, a diffuser and a heat exchanger to remove the heat of compression. The enriched UF6 product is withdrawn from one end of the cascade and the depleted UF6 is removed at the other end. The gas must be processed through some 1400 stages to obtain a product with a concentration of 3-4% U-235. Diffusion plants typically have a small amount of separation through one stage (hence the large number of stages) but are capable of handling large volumes of gas.
Commercial uranium enrichment was first carried out by the diffusion process in the USA, at Oak Ridge, Tennessee. The process was also used in Russia, UK, France, China and Argentina as well, but only on a significant scale in the USA and France in recent years. Russia phased out the process in 1992 and the last diffusion plant was USEC's Paducah facility, which shut down in mid-2013. It is very energy-intensive, requiring about 2400 kWh per SWU*. USEC said that electricity accounted for 70% of the production cost at Paducah, which was the last of three large plants in the USA originally developed for weapons programs and had a capacity of some 8 million SWU per year. It was used to enrich some high-assay tails before being finally shut down after 60 years' operation. At Tricastin, in southern France, a more modern diffusion plant with a capacity of 10.8 million kg SWU per year had been operating since 1979 (see photo below). This Georges Besse I plant could produce enough 3.7% enriched uranium a year to fuel some ninety 1000 MWe nuclear reactors. It was shut down in mid-2012, after 33 years' continuous operation. Its replacement (GB II, a centrifuge plant – see above) has commenced operation.
* It has been estimated that 7% of total US electricity demand was from enrichment plants at the height of the Cold War, when 90% U-235 was required, rather than the reactor grades of 3-4%for power generation.
In recent years the gaseous diffusion process had accounted for about 25% of world enrichment capacity. However, though they have proved durable and reliable, gaseous diffusion plants reached the end of their design life and the much more energy-efficient centrifuge enrichment technology has replaced them.

The large Georges Besse I enrichment plant at Tricastin in France (beyond cooling towers) was shut down in 2012.
Most of the output from the nuclear power plant (4x915MWe net) was used to power the enrichment facility.
Electromagnetic process
A very early endeavour was the electromagnetic isotope separation (EMIS) process using calutrons. This was developed in the early 1940s in the Manhattan Project to make the highly enriched uranium used in the Hiroshima bomb, but was abandoned soon afterwards. However, it reappeared as the main thrust of Iraq's clandestine uranium enrichment program for weapons discovered in 1992. EMIS uses the same principles as a mass spectrometer (albeit on a much larger scale). Ions of uranium-238 and uranium-235 are separated because they describe arcs of different radii when they move through a magnetic field. The process is very energy-intensive – about ten times that of diffusion.
Aerodynamic processes
Two aerodynamic processes were brought to demonstration stage around the 1970s. One is the jet nozzle process, with demonstration plant built in Brazil, and the other the Helikon vortex tube process developed in South Africa. Neither is in use now, though the latter is the forerunner of new R&D. They depend on a high-speed gas stream bearing the UF6 being made to turn through a very small radius, causing a pressure gradient similar to that in a centrifuge. The light fraction can be extracted towards the centre and the heavy fraction on the outside. Thousands of stages are required to produce enriched product for a reactor. Both processes are energy-intensive - over 3000 kWh/SWU. The Helikon Z-plant in the early 1980s was not commercially oriented and had less than 500,000 SWU/yr capacity. It required some 10,000 kWh/SWU.
The Aerodynamic Separation Process (ASP) being developed by Klydon in South Africa employs similar stationary-wall centrifuges with UF6 injected tangentially. It is based on Helikon but pending regulatory authorization it has not yet been tested on UF6 - only light isotopes such as silicon. However, extrapolating from results there it is expected to have an enrichment factor in each unit of 1.10 (cf 1.03 in Helikon) with about 500 kWh/SWU and development of it is aiming for 1.15 enrichment factor and less than 500 kWh/SWU. Projections give an enrichment cost under $100/SWU, with this split evenly among capital, operation and energy input.
One chemical process has been demonstrated to pilot plant stage but not used. The French Chemex process exploited a very slight difference in the two isotopes' propensity to change valency in oxidation/reduction, utilising aqueous (III valency) and organic (IV) phases.
Enrichment of reprocessed uranium
In some countries used fuel is reprocessed to recover its uranium and plutonium, and to reduce the final volume of high-level wastes. The plutonium is normally recycled promptly into mixed-oxide (MOX) fuel, by mixing it with depleted uranium.
Where uranium recovered from reprocessing used nuclear fuel (RepU) is to be re-used, it needs to be converted and re-enriched. This is complicated by the presence of impurities and two new isotopes in particular: U-232 and U-236, which are formed by or following neutron capture in the reactor, and increase with higher burn-up levels. U-232 is largely a decay product of Pu-236, and increases with storage time in used fuel, peaking at about ten years. Both decay much more rapidly than U-235 and U-238, and one of the daughter products of U-232 emits very strong gamma radiation, which means that shielding is necessary in any plant handling material with more than very small traces of it. U-236 is a neutron absorber which impedes the chain reaction, and means that a higher level of U-235 enrichment is required in the product to compensate. For the Dutch Borssele reactor which normally uses 4.4% enriched fuel, compensated enriched reprocessed uranium (c-ERU) is 4.6% enriched to compensate for U-236. Being lighter, both isotopes tend to concentrate in the enriched (rather than depleted) output, so reprocessed uranium which is re-enriched for fuel must be segregated from enriched fresh uranium. The presence of U-236 in particular means that most reprocessed uranium can be recycled only once - the main exception being in the UK with AGR fuel made from recycled Magnox uranium being reprocessed. U-234 is also present in RepU, but as an alpha emitter it does not pose extra problems. Traces of some fission products such as Tc-99 may also carry over.
All these considerations mean that only RepU from low-enriched, low-burnup used fuel is normally recycled directly through an enrichment plant. For instance, some 16,000 tonnes of RepU from Magnox reactors* in UK has been used to make about 1650 tonnes of enriched AGR fuel, via two enrichment plants. Much smaller quantities have been used elsewhere, in France and Japan. Some re-enrichment, e.g. for Swiss, German and Russian fuel, is actually done by blending RepU with HEU.
* Since Magnox fuel was not enriched in the first place, this is actually known as Magnox depleted uranium (MDU). It assayed about 0.4% U-235 and was converted to UF6, enriched to 0.7% at BNFL's Capenhurst diffusion plant and then to 2.6% to 3.4% at Urenco's centrifuge plant. Until the mid 1990s some 60% of all AGR fuel was made from MDU and it amounted to about 1650 tonnes of LEU. Recycling of MDU was discontinued in 1996 due to economic factors.
A laser process would theoretically be ideal for enriching RepU as it would ignore all but the desired U-235, but this remains to be demonstrated with reprocessed feed.
Tails from enriching reprocessed uranium remain the property of the enricher. Some recycled uranium has been enriched by Tenex at Seversk for Areva, under a 1991 ten-year contract covering about 500 tonnes UF6. French media reports in 2009 alleging that wastes from French nuclear power plants were stored at Seversk evidently refer to tails from this.
Enrichment of depleted uranium tails
Early enrichment activities often left depleted uranium tails with about 0.30% U-235, and there were tens of thousands of tonnes of these sitting around as the property of the enrichment companies. With the wind-down of military enrichment, particularly in Russia, there was a lot of spare capacity unused. Consequently, since the mid 1990s some of the highest-assay tails have been sent to Russia by Areva and Urenco for re-enrichment by Tenex. These arrangements however cease in 2010, though Tenex may continue to re-enrich Russian tails. Tenex now owns all the tails from that secondary re-enrichment, and they are said to comprise only about 0.10% U-235.
After enrichment
The enriched UF6 is converted to UO2 and made into fuel pellets – ultimately a sintered ceramic, which are encased in metal tubes to form fuel rods, typically up to four metres long. A number of fuel rods make up a fuel assembly, which is ready to be loaded into the nuclear reactor. See Fuel Fabrication paper.
Environmental Issues
With the minor exception of reprocessed uranium, enrichment involves only natural, long-lived radioactive materials; there is no formation of fission products or irradiation of materials, as in a reactor. Feed, product, and depleted material are all in the form of UF6, though the depleted uranium may be stored long-term as the more stable U3O8.
Uranium is only weakly radioactive, and its chemical toxicity – especially as UF6 – is more significant than its radiological toxicity. The protective measures required for an enrichment plant are therefore similar to those taken by other chemical industries concerned with the production of fluorinated chemicals.
Uranium hexafluoride forms a very corrosive material (HF – hydrofluoric acid) when exposed to moisture, therefore any leakage is undesirable. Hence:
- in almost all areas of a centrifuge plant the pressure of the UF6 gas is maintained below atmospheric pressure and thus any leakage could only result in an inward flow;
- double containment is provided for those few areas where higher pressures are required;
- effluent and venting gases are collected and appropriately treated.
Notes & References
General sources
Heriot, I.D. (1988). Uranium Enrichment by Centrifuge, Report EUR 11486, Commission of the European Communities, Brussels.
Kehoe, R.B. (2002). The Enriching Troika, a History of Urenco to the Year 2000. Urenco, Marlow UK.
Wilson, P.D. (ed)(1996). The Nuclear Fuel Cycle – from ore to wastes. Oxford University Press, Oxford UK.
IAEA 2007, Management of Reprocessed Uranium – current status and future prospects, Tecdoc 1529.